Hydrogen Sulfide (H2S) is a gas commonly found during the drilling and production of crude oil and natural gas, plus in wastewater treatment and utility facilities and sewers. The gas is produced as a result of the microbial breakdown of organic materials in the absence of oxygen. Colorless, flammable, poisonous and corrosive, H2S gas is noticeable by its rotten egg smell. With toxicity similar to carbon monoxide, which prevents cellular respiration, monitoring and early detection of H2S could mean the difference between life and death.

Hydrogen sulfide occurs naturally in some environments such as sulfur springs, swamps and salt marshes, and is often associated with the decomposition of organic material.
Human activities and industries that may produce hydrogen sulfide include:
● sewage treatment plants
● tanneries
● piggeries
● manure handling operations.
Scientists discussed a technology for cleaning natural reservoirs from hydrogen sulphide to improve the environment. The choice of research methods was to organize the creation of an airlift due to the electrolysis of seawater. To purify seawater from hydrogen sulphide, electrolysis with the release of hydrogen and oxygen is used. The released gases create an airlift effect, due to which the hydrogen sulphide rises to the surface, where it is captured for further separation. By-products of the electrolysis of seawater, chlorine and sodium, due to their good solubility in water, are converted into alkali and acid and, as a result of the neutralization reaction, return to the reservoir. Scientists also discuss the features of the electrolysis of seawater. The use of the proposed approach will improve the ecological situation and, in particular, will lead to an increase in fish stocks and in the restoration of the red algae population.
Hydrogen sulfide in the Black Sea was discovered in the late 19th century. And since then, there have been extensive discussions about its possible use. On the one hand, hydrogen sulfide is a renewable potential source of energy and sulfur. But, on the other hand, there is an environmental problem with the rise of hydrogen sulfide in the surface sea layers. The hydrogen sulfide zone begins at depths of 150-200 meters with a gradual increase in the concentration to 13-14 ml/l. But the relative concentration of hydrogen sulfide is small, which causes problems with the profitability of its production from large volumes of water with simultaneous purification from impurities. The concentration of hydrogen sulfide is a fraction of a percent relative to its saturated solution. Thus, the development and justification of cost-effective technologies for the use of hydrogen sulfide is currently relevant.
At various times, a number of approaches using airlift have been considered. In the early 90's, Ilya Varshavsky considered a technology in which it was supposed to create an airlift from hydrogen bubbles produced by the chemical reaction of activated aluminium with seawater. The hydrogen, rising up through the pipe, carried up the water along with the hydrogen sulphide dissolved in it. From the suspension moving to the surface of the water, due to the pressure drop, dissolved hydrogen sulphide began to be released, which enhanced the effect of the air lift. This technology used activated aluminium based on an alloy of aluminium with indium and gallium. Such an alloy successfully releases hydrogen from water, while indium and gallium are irretrievably consumed, which is economically unprofitable, since these components are close to gold in cost.
As an alternative, instead of indium and gallium, it is proposed to use an aluminium alloy with a few percent of copper, for example, 5-8% , or to use the alloy D16 (duralumin), which also contains copper. These alloys are well established as a cathode material in the electrolysis of water to produce hydrogen. In the proposed method, it is cheaper to organize an airlift using the decomposition of water by electrolysis. This work and experiments were conducted at the Mechanical Engineering Research Institute of the Russian Academy of Sciences. Figure 1 shows a diagram of a device that implements the proposed technology. The pipe is made of a light aluminium alloy AMG-6, almost not subject to corrosion in seawater. The pipe in this case is the anode, and the cathode is a round solid cylinder made of aluminium alloy located in the center. The lower end of the pipe is closed, and water with dissolved hydrogen sulphide enters through the side water intakes, which involves moving the pipe with the help of a vessel, i.e., water purification from hydrogen sulphide occurs in a decent water area. The closed lower part of the pipe involves the collection of electrochemical reaction products that are heavier than water and are not soluble in water. The advantage of this method is that this process will be regulated by changing the power of electricity supplied from a constant current source to the anode-cathode circuit. Unlike the technical solution, it will not be necessary to use a pump to organize the initial movement of water with hydrogen sulphide dissolved in it up to the surface of the reservoir. This is done by applying a large amount of power to the electrolysis cell at the beginning.
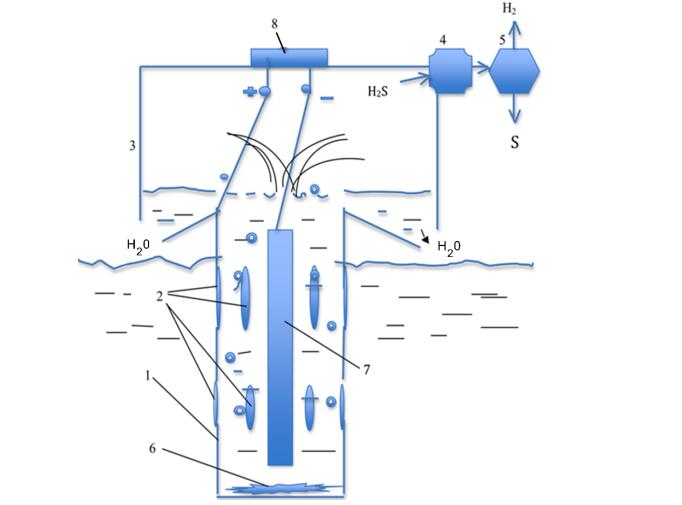
1-pipe-anode, 2-water intakes, 3-receiver, 4-compressor, 5-electrolyzer, 6-insoluble precipitate, 7-cathode.
Figure. Scheme of the installation for the purification of seawater from hydrogen sulphide.
TYHJ focuses on specialty gases development. We have full range of specialty gases, including electronic gases, high purity gases, mixed gases, electric lighting gases, ultra pure gases, laser gases, medical gases, calibration gases. As an ISO9001 and ISO14001 certified company, TYHJ exports gases into more than 30 countries and regions. We welcome your any inquiries. Please feel free to contact us.